Meet the Experts
Guest Editor

For many people, bowel cancer is diagnosed during a colonoscopy, where a small flexible camera is inserted into the rectum allowing a clinician to visually inspect the patient’s colon. If any abnormal growths are identified, these can be biopsied or removed during the procedure and the tissue is sent for assessment by a pathologist. For many years, people would be diagnosed with bowel cancer when they start to develop symptoms, however, the introduction of the bowel cancer screening test means that patients without symptoms can be identified early and prioritised for colonoscopy. Due to the widespread implementation of free national screening programmes, the predominant stage of bowel cancer diagnosis has recently shifted from late- to early-stage. However, worrying uptake of the bowel cancer screening test still remains at about 50%, meaning that almost half of the people that receive the test in Northern Ireland do not return it, which reduces their chances of being diagnosed early and having an improved outcome if they develop bowel cancer. Following diagnosis, identifying which bowel cancer patients are likely to respond to different types of treatment can be particularly challenging for clinicians. A number of clinical studies have indicated that a ‘one size fits all’ treatment approach for bowel cancer isn’t a viable option if we are to effectively tackle this disease.
In order to develop better treatments for individual patients, we must first understand the biology of that person’s tumour; this is the basis of personalised medicine in cancer. Advances in molecular and genetic analysis in the past 10 years have markedly improved our biological understanding of colorectal cancer (CRC), researchers within the Patrick G Johnston Centre for Cancer Research (PGJCCR) are using these state-of-the-art technologies to identify biomarkers that will help clinicians to select the most appropriate treatment options for cancer patients.
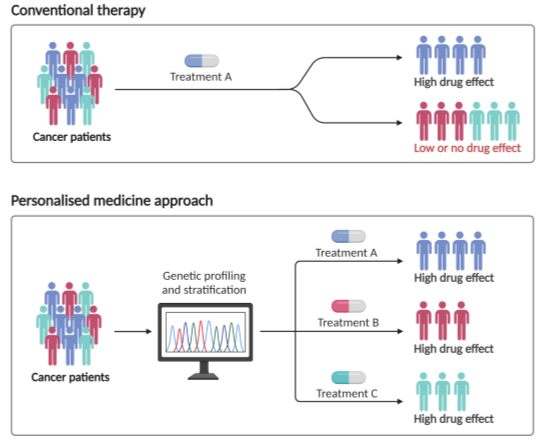
Through collaborations between QUB clinicians and scientists, our research is using new understanding developed in research laboratories, to identify the biology underlying an individual patient’s tumour and informing treatment decisions being made in the clinic; the “bench-to-bedside” approach. Our QUB research teams work with tumour samples that are removed during surgery, and work in our labs combines features from the tumour’s visual appearance down the microscope with information about changes in the genetic make-up of the person’s tumour. This approach is of particular relevance in recent years, as clinicians look to use molecular analysis to better understand the disease, which in turn enables the selection of the most appropriate drug for that patient’s individual tumour. The ultimate aim of this work is to allow patients to receive a more tailored disease management plan based on the specific properties and behaviour of their tumour. Thus, we can tailor treatment to the individual patient, maximising its effectiveness while minimising potential side effects.
The work ongoing in PGJCCR emphasises the need for the modern cancer researcher to have an interdisciplinary skillset; combining mechanistic biology, pathology and data analysis, in order to fully exploit and most accurately interpret data and their clinical implications. The research in our Belfast laboratories and clinical trial unit represents an excellent example of the best UK science and clinical care in bowel cancer working together to develop innovative approaches to save more lives.
The Dunne group have utilised this combined molecular pathology approach to identify a subset of patients with bowel cancer that have tumours distinguishable by features associated with the normal tissue surrounding the tumour itself, which have a particularly poor outcome after surgery. While this information is important to identify patient most at risk of disease relapse, the Dunne group are collaborating with researchers across the UK, Spain and Italy, as part of a Colorectal Cancer Stratified Medicine to test new treatment options for patients diagnosed with this particular tumour type. The group have recently published early results that indicate a type of immunotherapy that reduces relapse in laboratory-based testing and it is hoped may prove beneficial for these patients; these early findings will now move forward to be tested in a UK-based clinical trial within the next 5 years.
