Student Corner
Debbie Moss works in the lab of Dr Emma Kerr, which specialises in studies in cancer metabolism, and how cancer cells energetic demands and strategies for survival can be used to identify their weaknesses and develop more effective treatments. See below how she is exploiting this avenue to bring a refined approach to therapy.
Below, you will also find how maths and statistics are used in cancer research. Olivia Adair is a PhD student supervised by Dr Felicity Lamrock and is based at Mathematical Sciences Research Centre. Her project aims to understand and account for the cost of healthcare, and to refine strategies for sustainable and effective screening programmes for early detection of CRC.
OXIDATIVE MITOCHONDRIAL METABOLISM: A KEY DRIVER OF THERAPY RESISTANCE IN COLORECTAL CANCER (Debbie Moss)
My work focuses on colorectal cancer (CRC) which is the second largest cause of cancer-related deaths.  This is partly because not all patients respond well to chemotherapy treatment, meaning it’s less good at killing their cancer cells, and we call this “therapy resistance”. Unfortunately, we can’t yet tell exactly who those patients might be. Ultimately, our goal is to understand the processes that drive this therapy resistance, and target these processes with new drugs in patients who need them. That’s where my PhD comes in.
This is partly because not all patients respond well to chemotherapy treatment, meaning it’s less good at killing their cancer cells, and we call this “therapy resistance”. Unfortunately, we can’t yet tell exactly who those patients might be. Ultimately, our goal is to understand the processes that drive this therapy resistance, and target these processes with new drugs in patients who need them. That’s where my PhD comes in.
Metabolism is the collective term for all the biochemical reactions that happen in your body. These reactions combine what we eat and drink with the oxygen we breathe, and convert that into energy that fuels all the normal body processes. In cancer cells these processes are hijacked and hyperactivated, as cancer cells need more energy and building blocks to help them grow faster. We call this phenomenon “metabolic reprogramming”. In fact, the main chemotherapy drug for CRC patients called 5-fluorouracil (or 5FU) is an “antimetabolite” which means it mimics certain metabolites within the body. 5FU specifically mimics a molecule needed for making DNA so it interferes with CRC cell growth. But, not only does metabolic reprogramming help the cancer cells grow and divide much faster than normal cells, it also helps them survive stressful situations – like when they begin to move through the body, or when they’re treated with chemotherapy. So, metabolism can play an important role in resistance to 5FU chemotherapy for CRC patients.
We’ve discovered that patients whose cancers can make lots of energy are more resistant to 5FU-chemotherapy, and as part of my PhD I’ve been looking for drugs that might combine with 5FU to work better in killing CRC. Specifically, I’ve used drugs that target a core energy-producing metabolic pathway, called Oxidative Phosphorylation (OxPhos). I found that drugs which block the first step of that process are particularly effective in improving CRC cell response to 5FU.
In fact, we’ve discovered we can use the anti-diabetic drug Metformin to do just that, blocking CRC cell growth and triggering more CRC cell death when we give it in combination with 5FU-chemotherapy.
Since Metformin is safe, and can already be given to CRC patients when on chemotherapy treatment (like for diabetic CRC patients) we hope we can begin to make a difference to those patients who desperately need new options for treatment.
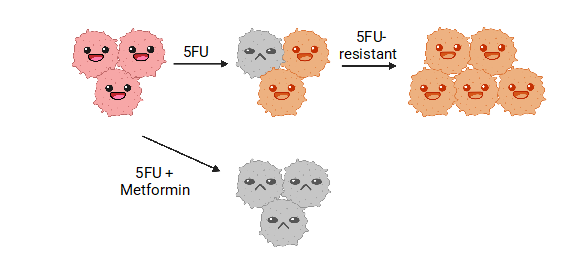
5FU is effective on some bowel cancer cells, but some develop resistance and continue to grow in its presence; by adding metformin to a typical 5FU treatment, most cancer cells will not survive, showing the combination of this safe drug to our typical standard of care can be used to improve effectiveness.
My PhD focuses on cancer prevention (Olivia Adair).
 This type of cancer starts in the large intestine, also known as colon cancer (or colorectal cancer or bowel cancer). This type of cancer can start as small lumps that grown insider the colon where they shouldn’t. These lumps are small tumours, also known as polyps. Most polyps aren’t cancer but can turn into cancer if their size increases. Polyps less than 1 cm in size have just over a 1% chance of becoming cancer and polyps greater than 2 cm in size have a 40% chance of becoming cancer. In general, 5% of polyps turn into cancer, so it is usually better to remove any polyps before they turn into cancer.
This type of cancer starts in the large intestine, also known as colon cancer (or colorectal cancer or bowel cancer). This type of cancer can start as small lumps that grown insider the colon where they shouldn’t. These lumps are small tumours, also known as polyps. Most polyps aren’t cancer but can turn into cancer if their size increases. Polyps less than 1 cm in size have just over a 1% chance of becoming cancer and polyps greater than 2 cm in size have a 40% chance of becoming cancer. In general, 5% of polyps turn into cancer, so it is usually better to remove any polyps before they turn into cancer.
This can be done by having a look into the colon with a small camera on the end of a long tube (colonoscopy), as people with polyps don’t know they have them as they feel well.
In order to decide who should get a colonoscopy, some tests are performed first. By collecting your stool sample and having the hospital perform a test on it, doctors can detect if there might be a problem. This process is known as screening.
Of all the people who have colon cancer, some don’t feel well and turn up at the hospital or their doctor’s office, and some people are screened and find out they have cancer that way. 
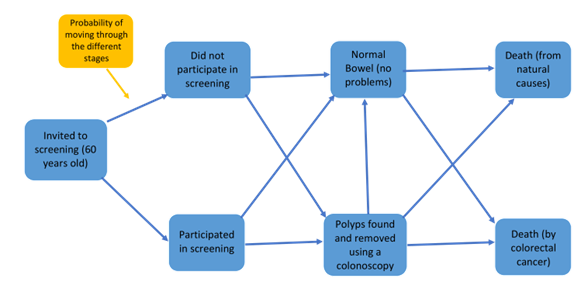
Screening in Northern Ireland is managed by the Bowel Cancer Screening Programme and was set up in April 2010. Screening is helpful to find polyps early and remove them to prevent progression to cancer, providing an opportunity for an earlier diagnosis to help those patients live longer. Screening is typically offered to people aged between 60 and 70 years old and if all looks okay, they will be screened again every 2 years.
The more people are screened early, the better chance of removal and effective treatment, but screening everyone would cost a lot of time and money.
My PhD project is to build a mathematical model which looks at the screening programme in Northern Ireland to see when it is good value for money to perform these tests, and investigate what screening approach is the most cost effective for Northern Ireland.