School Research Seminars

Each academic year, the School of Chemistry and Chemical Engineering hosts a diverse range of research academics from institutions across the globe, providing the opportunity for discussion, collaboration, and idea sharing in relation to cutting-edge research within the chemical sciences.
Upcoming School research seminars are detailed below - should you wish to attend one of the seminars, please contact our School Seminar Coordinator.

For the last number of years, ours and other research groups have been looking at ways to undertake continuous monitoring of health in non-invasive ways. While some exciting innovations in this field related to continuous or semi-continuous biomarker monitoring in sweat and interstitial fluid has been reported, our approach has focussed on capturing and analysing the volatile compounds that are coming from our skin.
The skin surface emits many classes of volatile organic compounds (VOCs), as well as ammonia, which are derived directly from glandular secretions and also from their interactions with resident skin bacteria. These VOC emissions have the potential to offer insights into cutaneous and systemic physiology. However, in general, and in contrast to breath volatile research, research into skin-emitted VOCs as they relate to pathological state is at an early stage. Skin volatile profiles of healthy individuals have been shown to be influenced by gender and other genetic traits. However, little consistency has been seen across studies with regards to the compounds observed, likely due to the wide variety of sampling and analysis approaches being used but also the highly variable nature of the skin matrix itself. More research is required to more deeply understand the healthy skin volatile profile and the impact of disease on this emission.
This talk will cover the work that we have contributed to understanding the skin volatile emission using gas chromatography-mass spectrometry, how we are translating what we know for future wearable devices and what we believe is required of the field in order to exploit our knowledge and methodologies for clinical volatilomics in disease monitoring. Specifically, we are interested in skin volatilomics in the context of skin barrier disruption and also clinical wound volatilomics for pathogen detection. I will discuss our work to date and our perspectives on monitoring the skin volatile emission as a potential diagnostic tool for addressing needs in these specific application areas.

Harmful blue-green algae (cyanobacteria) can pose significant challenges to drinking water treatment with instances of increased biomass and harmful secondary metabolites in the raw water. Conventional water treatment processes can often be challenged and even overwhelmed by harmful algal blooms (HABs).
Advanced oxidation processes in general and semiconductor photocatalysis particularly can represent a solution to the challenges posed by HABs. With an iterative approach to designing and testing a novel UV-LED TiO2-based treatment system we were able improve the raw water quality in a Brazilian reservoir in a pilot scale study.

Ionic liquids (ILs) have attracted significant interest as replacement solvents in electrochemical experiments over the last two decades. They possess several properties make them attractive compared to traditional solvent/electrolyte combinations, including high chemical and thermal stability, wide electrochemical windows, low volatility, high polarity, high viscosity and the ability to dissolve a wide range of compounds. My group and I are interested in employing ionic liquids with low-cost planar electrode devices for the detection of target analytes including gases (e.g. oxygen, hydrogen, ammonia, sulfur dioxide, chlorine, methylamine) and explosive compounds (e.g. 2,4,6-trinitrotoluene (TNT), hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX), and octahydro-1,3,5,7-tetranitro- 1,3,5,7-tetrazocine (HMX)).
In this presentation, Dr Silvester-Dean will discuss some of her recent developments in this area, focussing on: (1) the sensing of gases in ILs with different humidity environments, revealing that ion structuring at the electrode is disrupted in the presence of moisture, (2) mixing with polymers to physically stabilise the IL on the electrode, a move towards robust wearable sensors, and (3) the preconcentration ability of ionic liquids that allows the detection of explosive compounds such as TNT using liquid/liquid partitioning. Our long-term goal is to develop a new generation of low-cost commercially available sensors comprising of ionic liquids or gelled electrolytes, for the fast and reliable detection of analytes.
---
Assoc. Prof. Debbie Silvester is an electrochemist and ARC Future Fellow in the School of Molecular and Life Sciences at Curtin University, Perth. She completed her DPhil (PhD) at the University of Oxford, UK, then spent a short time as an intern for Schumberger Cambridge Research, before arriving at Curtin University as a Curtin Research Fellow. In 2012, she was awarded an ARC Discovery Early Career Research Award (DECRA) and in 2017, an ARC Future Fellowship.
Her research focusses on the application of ionic liquids as electrolytes in electrochemical reactions, with an emphasis on understanding fundamental behaviour to develop new electrochemical sensors. She has won various awards for her research, including a Young Tall Poppy award, the Rennie Memorial medal from the Royal Australian Chemical Institute, the Peter W. Alexander Medal from the Analytical & Environmental Division of the RACI, and the Alan M Bond medal from the Electrochemistry Division of the RACI. She is currently the secretary for the Electrochemistry Division of the RACI, and the Australia/New Zealand representative for the International Society of Electrochemistry.

Elucidating Proton-Coupled Electron Transfer Mechanisms Underpinning the Catalytic Generation of Renewable Fuels
The conversion of energy-poor feedstocks like water and carbon dioxide into energy-rich fuels involves multi-electron, multi-proton transformations. In order to develop catalysts that can mediate fuel production with optimum energy efficiency, this complex proton-electron reactivity must be carefully considered.
Using a combination of electrochemical methods and time-resolved spectroscopy, we have revealed new details of how molecular catalysts mediate the reduction of protons to dihydrogen and the experimental parameters that dictate catalyst kinetics and mechanism. Through these studies, we are revealing opportunities to promote, control and modulate the proton-coupled electron transfer reaction pathways of catalysts.

Sabatier’s principle,[1] developed in the first decades of the 20th century, states that an ideal catalyst should bind a substrate neither too strongly nor too weakly. Today, this simple idea provides the fundamental underpinning for “volcano plots”,[2,3] which are abundantly used in heterogeneous and electrocatalysis.[4]
Recently, Prof. Corminboeouf's group have elaborated a computational toolkit[5] based on Sabatier's principle that facilitates the identification of overarching trends present in the “catalysis space” and provides new insights in the best places to “look” for homogeneous catalysts having high activity.
For a given reaction, prospective species anticipated to exhibit enhanced activity can be identified and accompanying design principles that should be useful for synthetic chemists constructed. The combination of these tools with supervised machine-learning techniques enable the expansion of the pool of examined catalysts to over 105 species.
The large quantity of data generated can be compiled and mined into an interactive tool, which facilitates the analysis and assists in identifying the most compatible metal/ligand family combinations with the goal of establishing relationships between the intrinsic chemical properties of different catalysts and their overall catalytic performance. Prof. Corminboeouf's group focus on both prototypical classes reactions and challenging organic processes and highlight some of the pro and cons of the overall approach.
References
[1] P. Sabatier, Ber. Dtsch. Chem. Ges. 1911, 44, 1984.
[2] H. Gerischer, Bull. Soc. Chim. Belg. 1958, 65, 506.
[3] R. Parsons, Trans. Faraday Soc. 1958, 54, 1053.
[3] J. K. Nørskov, T. Bligaard, J. Rossmeisl, C. H. Christensen, Nat. Chem. 2009, 1, 37.
[4] M. D. Wodrich, B. Sawatrlon, M. Busch, , C. Corminboeuf, Acc. Chem. Res. 2021, 6, 6754.

Protic ionic liquids (PILs) are cost efficient “designer” solvents which can be tailored to have properties suitable for a broad range of applications1-2. More recently ionic liquids (ILs) are being combined with molecular solvents, and these binary or multicomponent IL solvents enables more control over the solvent environment, and typically reduces the cost and viscosity.
However, there are relatively few structure-property studies which look at these more complex mixtures. We have explored the solvation properties of common PIL-molecular solvents using various techniques, including Kamlet-Aboud-Taft parameters determined from solvatochromic dyes, and MD simulations3-4. These have identified many interesting solvent properties of these solutions and gives insight into their interactions with solutes. This lead to the use of PIL-water solvents having the potential to control protein solubility and stability, which in turn are critical for applications in bioprocessing, protein crystallography and cryopreservation. In this presentation I will cover some solvation properties of PIL solvents, along with the protein structure changes of proteins in IL solvents which we have explored using spectroscopic and advanced small angle x-ray scattering (SAXS) methods5.
References
1. Greaves, T. L.; Drummond, C. J., Protic ionic liquids: properties and applications. Chem. Rev. 2008, 108 (1), 206-237.
2. Greaves, T. L.; Drummond, C. J., Protic ionic liquids: evolving structure−property relationships and expanding applications. Chem. Rev. 2015, 115, 11379-11448.
3. Yalcin, D.; Christofferson, A. J.; Drummond, C. J.; Greaves, T. L., Solvation properties of protic ionic liquid – molecular solvent mixtures. Phys. Chem. Chem. Phys. 2020, 22, 10995-11011.
4. Yalcin, D.; Drummond, C. J.; Greaves, T. L., Solvation properties of protic ionic liquids and molecular solvents. Phys. Chem. Chem. Phys. 2020, 22, 114-128.
5. Han, Q.; Smith, K. M.; Darmanin, C.; Ryan, T. M.; Drummond, C. J.; Greaves, T. L., Lysozyme conformational changes with ionic liquids: Spectroscopic, small angle x-ray scattering and crystallographic study. Journal of Colloid & Interface Science 2020, DOI: 10.1016/j.jcis.2020.10.024.

About a century ago, Dr. Hermann Staudinger substantiated the existence of ultralong molecules and won the longterm debate against the colloidal theory to establish polymer science. Needless to say, polymer science has made tremendous contributions to the progress of human society, although it coincidentally brought about a critical environmental issue to tackle.
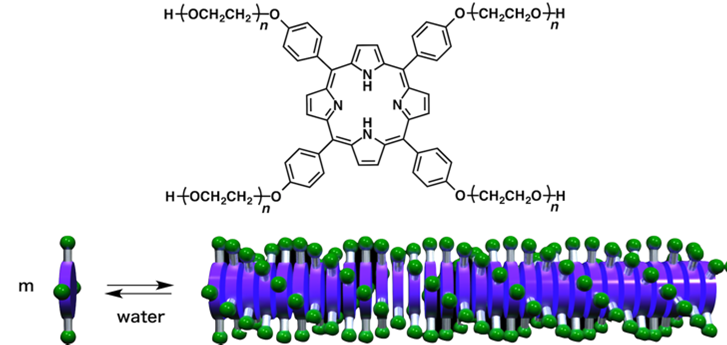
In his talk, Professor Aida will present the significance and applications of supramolecular polymerization, a modernized version of the colloidal approach to polymeric materials. Supramolecular polymers attract attention not only because they are 100% recyclable but also they can be designed to be environmentally friendly, self-healable, responsive, and/or adaptive [1–4]. In 1988, Prof. Aida and his team reported the first prototype of supramolecular polymerization, featuring the formation of a 1D polymeric assembly using an amphiphilic porphyrin with water-soluble oligoether side chains as the monomer and have made fundamental contributions to this field. Representative examples include (1) nanotubular upramolecular polymerization, (2) chain-growth supramolecular polymerization, (3) supramolecular block copolymerization, (4) stereoselective supramolecular polymerization, and (5) thermally bisignate supramolecular polymerization. These contributions are integral elements of conventional polymer science, filling in the critical gap between supramolecular and conventional polymerizations. Furthermore, Prof. Aida's team have expanded the basic concept of supramolecular polymerization into the noncovalent design of innovative soft materials. Successful examples include the developments of (i) bucky gels, (ii) aquamaterials, (iii) mechanically robust self-healable materials, (iv) supramolecular polymers of biomolecular machines, (v) ferroelectric columnar liquid crystals, and (vi) reorganizable and adaptive core-shell columnar liquid crystals. Prof. Aida will highlight some of these examples to show the significance of supramolecular polymerization for the realization of sustainable society.
REFERENCES
[1] Aida, Meijer, Stupp, Science 2012, 335, 813–817.
[2] Aida, Meijer, Israel J. Chem. 2020, 60, 33–47.
[3] Aida, Adv. Matter. Essay, 2020, 1908140.
[4] Hashim, Bergueiro, Meijer, Aida, Prog. Polym. Sci. 2020, 101250.

Converting simple starting materials into complex products using only a light source (synthetic photochemistry) is especially attractive to organic chemists, particularly from the point of view of green chemistry: waste is minimized, and light is readily available. In addition, photochemical routes often allow efficient access to complex frameworks (particularly to strained molecules and intermediates) that cannot be generated using ground-state chemistry.
In this seminar, Dr Coote's recent work on the synthesis and applications of bicyclic 1,2-diazetidines2 will be presented. Bicycles2 can be obtained in high yields from 1,2-dihydropyridazines1 simply upon irradiation at 350 nm, and are versatile synthetic intermediates that can be converted into a variety of different derivatives, including substituted 1,2-diazetidines, cyclobutanes, cyclobutenes and dienes (Figure 1).1,2,3 In addition, related work on similar systems will be discussed, including ongoing unpublished work.
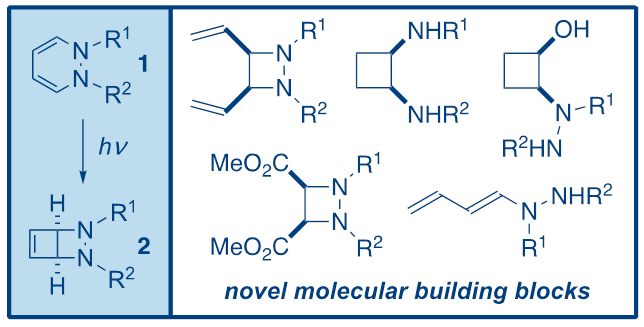
Figure 1.
References
1. Britten, T. K.; Kemmitt, P. D.; Halcovitch, N. R.; Coote, S. C. Org. Lett. 2019, 22, 9232
2. Britten, T. K.; Akien, G. R.; Kemmitt, P. D.; Halcovitch, N. R.; Coote, S. C. Tetrahedron Lett. 2019, 60, 1497
3. Britten, T. K.; Kemmitt, P. D.; Halcovitch, N. R.; Coote, S. C. Synlett 2020, 31, 459
