The LifeArc Centre for Acceleration of Rare Disease Trials
Queen's University of Belfast co-lead the LifeArc Centre for Acceleration of Rare Disease Trials with University of Birmingham and Newcastle University:
- Globally, there are more than 450 million people living with rare diseases; >110,000 people across Northern Ireland.
- 75% of rare diseases start in childhood with 30% of affected children dying before their 5th birthday.
- For most people living with rare disease there is no cure.
- <5% of rare diseases have an approved therapy
- People living in Northern Ireland with rare diseases do not have equity of access to diagnosis, therapy, or research.
Clinical trials for rare diseases are challenging and can be a major limiting step in getting new treatments to patients. The research can be fragmented, and researchers sometimes lack access to specialist facilities, as well as advice on maximising trial recruitment and keeping people within trials, managing data and trial regulations, optimising trial designs, and translational project management. Our LifeArc Centre for Acceleration of Rare Disease Trials brings together a consortium of three universities from across the UK to boost the capacity and efficiency of rare disease trials across the UK. Queen’s University Belfast, Newcastle University, and University of Birmingham are pooling their expertise in a partnership with a 4-nation approach. This £12M Centre will focus on helping patients have access to appropriate treatments earlier, delivering trials of new treatments using ‘one stop’, patient friendly, models and facilitating equitable recruitment. We will speed up the delivery of clinical trials for people with rare diseases and enable more rapid approval of new therapies for use in the NHS. We are working alongside people living with rare disease(s) and their families to ensure 'nothing about us without us' trial designs and delivery, ensuring the right information is collected that improves patient and / or family life, and enabling treatments to be approved more quickly. People affected by rare disease are involved at every stage in this project from the initial idea, designing the project, as formal investigators on the funding application, leading individual elements, and helping deliver the LifeArc Centre for the Acceleration of Rare Disease Trials. We have a broad outreach and are keen to welcome participation and involvement throughout.
More about The LifeArc Acceleration of Rare Disease Trials at QUB:
What does this mean for Northern Ireland?
We know that Northern Ireland is underrepresented in clinical trials, in clinical research with children, for rare disease research funding, and for rare disease clinical centres of expertise. This project, and other complementary research, will help improve research opportunities and facilitate access to treatment for people living with rare diseases.
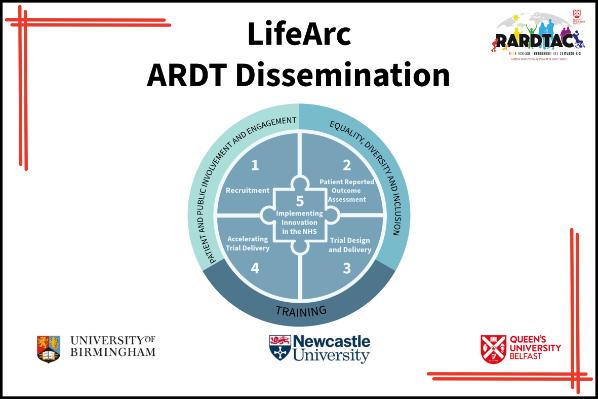
Work package 1:
- We are developing a consent register for Northern Ireland where people can sign up to be kept informed about various research opportunities.
- We are developing a secure online recruitment portal where people living with rare diseases can self-register their interest in clinical trials, linked to a clinical trial finder. Effectively, this will help link patients with clinical trials for which they may be eligible.
- We are developing core datasets, standardised data dictionaries that improve coding of rare diseases in electronic care records, and e-cohorts to support pre-screening of patients, collect 'real world' natural history data to inform patient reported outcomes, and enabling the use of innovative trial designs, patient simulation with digital twins, synthetic controls for innovative trial designs, and more personalised medicine. NI has an exceptional opportunity to lead the way using national population-based datasets with validated, quality controlled information for rare diseases.
Work package 2:
- We will develop a standardised approach to patient reported outcomes for rare disease trials, including developing consensus guidelines that support equity for trials
- We will identify what prevents inclusive collection of patient reports outcomes and explore how better approaches can be used / developed
- We will develop a bespoke design service for patient reported outcomes
Work package 3:
- Focuses on creating rare disease clinical trial capacity to support delivery of a portfolio of trials, integrating innovative trial designs, and delivering trials at scale. Newcastle and Birmingham have more impact in this WP as they already have established rare disease centres of excellence and rare disease trial units...Belfast is scaling up!
Work package 4:
- Will set up a rare disease trials network across Scotland, England, Wales, and Northern Ireland.
- We will work with NHS colleagues to develop a system so that one approval can be supported by multiple recruiting centres.
- In NI we will also set up an active rare disease clinical-academic research network, informing and enabling clinical trials
- We are putting in place core staff to help get local patients involved in clinical trials locally and / or overseas. A patient logistics and experience officer will support individual patients and their families from initial consent, through their trial journey, to completion of the trial.
- We are also working to de-centralise trials - enabling home health monitoring and / or recruitment supported by GPs
Work package 5:
- Supports implementation of rare disease trial innovations in the NHS, developing new approaches and learning from international experts what works, when.
- We will conduct health economic analyses, constructing a Value of Information (VoI) framework and developing a standardised approach to patient-informed risk benefit trade-offs through Multiple Criteria Decision Analysis (MCDA)
- We will identify barriers to participation in clinical trials for our rare disease community, working with stakeholders to identify solutions
- We will identify regulatory barrier to accessing / sharing data and for approvals / access to treatment across the UK
- A Citizen's jury will address patient-relevant issues around early access to medicines
- We will work with the UK RD_IMAG group and international collaborators to maximise benefits for our rare disease community.
There are dedicated workpackages for patient involvement and engagement, as well as for equity, diversity and inclusion, ensuring a collaborative and equal contribution across all research. We are also helping build capacity for rare diseases across Northern Ireland, with a dedicated rare disease doctoral training program that will train the next generation of laboratory, computational, and clinical, researchers.


-600x399.jpeg)
