Dynamic Regulatory Alignment and the Protocol on Ireland/Northern Ireland - Two Year Review
Lisa Claire Whitten
February 2023
Download a copy of this Explainer here
Introduction
Northern Ireland occupies a unique position in the relationship between the United Kingdom (UK) and the European Union (EU). The Protocol on Ireland/Northern Ireland provides that aspects of EU law continue to apply in Northern Ireland despite it having left the EU with the rest of the UK on 31 January 2020.
Under the terms of the Protocol, Northern Ireland remains part of the UK customs territory. However, the EU customs code applies in respect of Northern Ireland as do specific EU acts that regulate certain individual rights, free movement of goods, VAT and excise, state aid and electricity markets. New EU acts that fall within the scope of the Protocol may also be added to those that apply in Northern Ireland.
Moreover, the Protocol requires that amendments or replacements to these acts apply in Northern Ireland. Such dynamic regulatory alignment is necessary to maintain the free movement of goods on the island of Ireland. However, this has proved politically controversial, not least because it involves EU acts applying in Northern Ireland in which, after Brexit, neither the UK nor Northern Ireland has had a direct role in adopting.
In June 2022, the UK government proposed domestic legislation – the Northern Ireland Protocol Bill (NIPB) – that would, if enacted, disapply those provisions of the Protocol in UK law that currently provide for the continued application of EU law in Northern Ireland. As the NIPB is still going through the parliamentary process the arrangements for Northern Ireland agreed to by the UK and the EU in the text of the Protocol still apply. This would change if the NIPB is adopted.
Focusing on the current legal situation, this explainer reviews the substance of the first two years of dynamic regulatory alignment’ with those elements of EU law applicable in post-Brexit Northern Ireland. The content builds on three previous reviews carried out after the first six months, 12 months and 18 months following the Protocol’s entry into force.
The extent of change after 24 months does not differ much from that presented after 18 months of the Protocol’s implementation, nor indeed from that presented after 12 months; this is itself an important finding. The absence of significant change throughout 2021 and 2022 reflects the slow pace of the EU legislative process and the relative stability of the specific set of EU acts that continue to apply in the UK in respect of Northern Ireland.
Regular minor amendments and technical updates to EU implementing legislation also apply to Northern Ireland. While many have little or no impact in Northern Ireland in terms of policy, some do have implications for industry and stakeholders with some changes also requiring dedicated domestic law to implement them. The wide variation in terms of policy impact of – sometimes frequent – amendments and updates to applicable EU law underlines the ongoing importance of monitoring the legal and practical implications of the unique position of Northern Ireland post-Brexit.
Given the generally slow pace of change, the policy impacts of dynamic regulatory alignment for Northern Ireland under the Protocol are not as extensive as they could be. This is not to say dynamic regulatory alignment is without its challenges in respect of the practical impacts for industry, democratic accountability, and legislative scrutiny. Following a review of the first two years of implementation, the conclusion to this explainer returns to these challenges.
1. The new dynamism of Northern Ireland
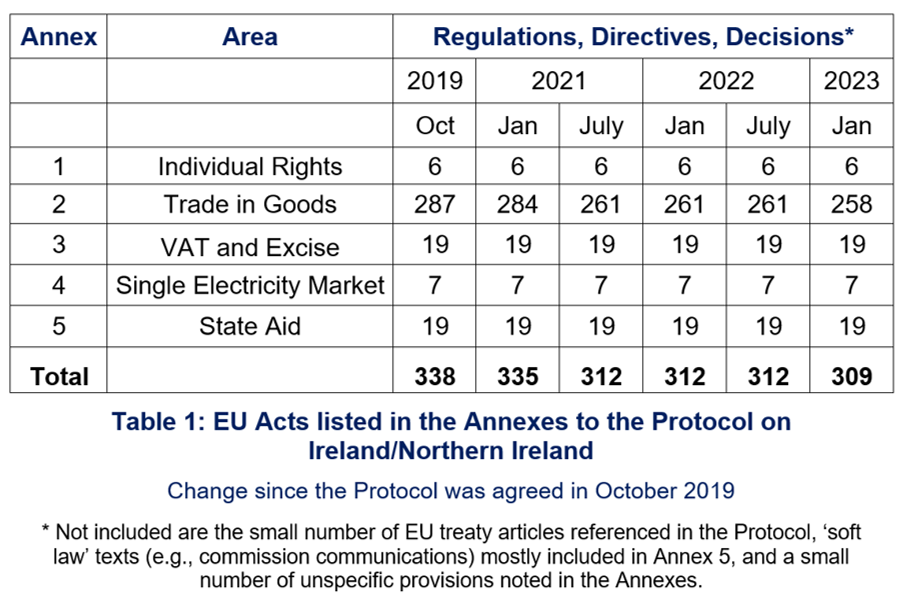
Under Article 13(3) Protocol, EU acts listed in Annexes to the Protocol apply ‘as amended or replaced’ to the UK in respect of Northern Ireland. When the Protocol and its Annexes were agreed by UK and EU negotiators as part of the UK-EU Withdrawal Agreement in October 2019, 338 acts were listed. Under the Protocol, additions can be made, and acts can also be deleted.
This means that, under the Protocol, Northern Ireland is in a position of ‘dynamic alignment’ with a specified but potentially evolving selection of the EU ‘acquis’, the body of legal and other agreed obligations and commitments that apply to, and in, EU member states.
In implementing the Protocol, therefore, the UK has committed to keeping Northern Ireland aligned with changes made to the EU acts included in the scope of the Protocol.
Two years after the Protocol entered into force, how has the body of EU law that applies to, and in, Northern Ireland changed?
Like many issues in the world of Brexit, the answer is not simple. Several types of change have taken place. They fall into four broad categories:
-
- additions to and deletions from the Annexes to the Protocol
- repeal, replacement, and expiry of applicable EU law
- amendments to applicable EU law
- changes to EU legislation that implements applicable EU law.
2. Additions to and deletions from the Annexes to the Protocol
The first category of change concerns the specific EU acts that apply in Northern Ireland under the Protocol. Through the Joint Committee set up to oversee the implementation of the UK-EU Withdrawal Agreement, the UK and the EU can, by agreement, add new EU acts that fall within the scope of the Protocol to the relevant Annexes (Article 13(4) Protocol). The Joint Committee can also remove acts listed.
Before the end of the 11-month transition period that followed the UK’s withdrawal from the EU, the UK and EU agreed in December 2020 to add eight EU acts to Annex 2 of the Protocol. It also agreed to remove two EU acts listed in the same Annex.
Of the eight acts added, five related to legislation that the Joint Committee decided, following review, should have been included in the original text of Annex 2. The five acts that were added concern:
-
- rules for monitoring trade between the EU and third countries in drug precursors (Council Regulation (EC) 111/2005);
- use of indications or marks to identify the lot – or batch – to which food products belong (Directive 2011/401/EU);
- rules on the marketing of fodder plant seed (Council Directive 66/401/EEC);
- rules on the marketing of propagating material of ornamental plants (Council Directive 98/56/EC); and
- rules on the marketing of vegetable propagating and planting material other than seed (Council Directive 2008/72/EC).
The three other additions were new EU acts adopted since the content of the Protocol had initially been agreed in November 2018. The Joint Committee decided that the following three acts fell within the scope of the Protocol, so added these to Annex 2:
-
- bilateral safeguard clauses and other mechanisms for the temporary withdrawal of preferences in certain EU trade agreements with third countries (Regulation (EU) 2019/287);
- measures to reduce the impact of certain plastic products on the environment (Directive (EU) 2019/904); and
- and measures to control the introduction and import of cultural goods (Regulation (EU) 2019/880).
The two acts that were removed by the Joint Committee concerned CO2 emissions standards for passenger cars (Regulation (EC) 443/2009) and light-duty commercial vehicles (Regulation (EU) 510/2011). Their original inclusion was deemed unnecessary.
Taking these changes into account, when the Protocol entered into force on 1 January 2021 following the end of the transition period, 344 EU acts were listed in its Annexes.
Although the Joint Committee has met on three occasions since then – 14 February 2021, 9 June 2021, and 21 February 2022 – it has not adopted any decision to add or delete any EU acts. The European Commission has nevertheless signalled that certain upcoming EU legislation may fall, in part at least, within the scope of the Protocol. This includes, for example, the proposed Carbon Border Adjustment Mechanism (CBAM) regulation which has been provisionally agreed by the EU. UK and EU officials have discussed its potential application to Northern Ireland but no definitive position has yet been taken. Another example is the EU’s ‘Geographical Indication’ (GI) rules covering craft and industrial products which the European Commission have a mandate to expand. The EU has informed the UK government that it would seek to add the new GI regulation to the Protocol under Article 13(4). No definitive position has yet been reached on this matter; the UK government has said it will ‘continue to monitor the situation’ regarding the new GI Regulation and the Protocol throughout the EU legislative process.[1]
3. Repeal, replacement, and expiry of applicable EU Law
The second category of change covers the repeal, replacement, and expiry of EU acts – regulations, directives, and decisions – listed in the Annexes to the Protocol. Changes in this category are the result of normal EU legislative processes and follow from the provision in Article 13(3) Protocol stating that relevant EU acts apply as ‘amended or replaced’ to and in Northern Ireland.
Of the 338 EU acts originally listed in the Annexes, 57 had been repealed as of 1 January 2023. Only five of these had been repealed in the preceding six months.
Most, but not all, of the repealed EU acts have been directly replaced by a new piece of EU legislation. This is because several replacement acts can consolidate provisions previously spread over various pieces of (now repealed) legislation into one or two new, more comprehensive, acts. The 57 repealed acts have been replaced by 23 new acts.
In most instances, even three years after the UK withdrew from the EU, this dynamic alignment concerns changes to pieces of EU legislation adopted when the UK was in the EU. Of the 23 replacement acts, only five were adopted after the UK left the EU on 31 January 2020; two in 2020 (Decision (EU) 2020/263 and Regulation 2020/740) – two in 2021 (Directive (EU) 2021/555 and Regulation (EU) 2021/821), and one in 2022 (Commission Regulation (EU) 2022/2473) – so after the end of the UK Transition Period; details about content and date of adoption are listed below.
In terms of coverage, 23 of the 57 repealed acts concerned controls on animal health and were replaced by two new pieces of legislation: Regulation (EU) 2016/429 and Commission Delegated Regulation (EU) 2020/687. The first of these is known as the ‘Animal Health Law’ and the second is a related, supplementary act. Together these two new acts incorporate and update pre-existing provisions set out in the 23 repealed acts.
The changes laid down in the Animal Health Law were agreed in March 2016, so before the UK’s EU referendum and therefore with the UK taking full part in their adoption. The adopted text included transitional measures and allowed for the repeal of earlier acts to take effect in April 2021.
As a supplement to the 2016 Regulation, the Commission Delegated Regulation (EU) 2020/687 sets out measures to prevent and control the spread of certain diseases. The relevant diseases were listed in the 2016 regulation but required more specific provisions; these are laid down in the Delegated Regulation.
In a similar way, seven of the other repealed acts concerned EU rules on official controls and checks on food and feed, animal health and welfare standards, plant health and plant protection. These were replaced by a single overarching EU act: Regulation (EU) 2017/625, known as the ‘Official Controls Regulation’. It incorporates and updates pre-existing provisions in the repealed acts. It was agreed in April 2017, shortly after the UK triggered Article 50 announcing formally its intention to withdraw from the EU and so with the UK participating in the regulation’s adoption. The new act (Regulation (EU) 2017/625) included transitional measures and allowed for the repeal of the seven earlier acts to take effect in December 2019.
Also repealed were two directives – Council Directive 93/42/EEC and Council Directive 90/385/EEC – concerning the production of and trade in medical devices. This had been provided for in Regulation (EU) 2017/745 which was already listed in Annex 2 to the Protocol, so the repealed directives were not replaced directly.
In addition, two regulations concerning requirements for the use of statistics on trade in goods between EU member states and with non-EU countries – Regulation (EC) No 638/2004 and Regulation (EC) No 471/2009 – were repealed and replaced by Regulation (EU) 2019/2152 on European business statistics. It incorporates and updates requirements from the earlier acts. The new regulation was agreed in November 2019, when the UK was still an EU member state; it also included transitional measures for the scheduled repeal of earlier acts to take effect at the end of 2021.
Also giving effect to a regulation adopted in November 2019, three regulations concerning the requirements for approving motor vehicles for use – Regulation (EC) No 78/2009, Regulation (EC) No 661/2009 and Regulation (EC) No 79/2009 – were repealed and replaced by Regulation (EU) 2019/2144 on requirements regarding general safety and protection of occupants applied to motor vehicles as well as to trailers, systems, components, and technical units used in them. This repeal and replacement came into effect in July 2022.
A further 19 repealed regulations and directives originally listed in the Protocol have been replaced directly by new acts. Of these replacement acts, four concern the regulation of electricity markets and energy supplies (Directive 2009/72/EC, Regulation (EC) 714/2009, Regulation (EC) 713/2009 and Directive 2005/89/EC) and were originally listed in Annex 4, supplementing Article 9 Protocol which makes provision for the continued operation of the Single Electricity Market on the island of Ireland. These four acts were replaced by four updated acts (Directive (EU) 2019/944, Regulation (EU) 2019/943, Regulation (EU) 2019/942 and Regulation (EU) 2019/941 respectively) between July 2019 and December 2020. The replacement acts cover the same policy areas and implement changes agreed in June 2019 – again while the UK was still a member state of the EU.
Of the remaining acts, 15 have been repealed and replaced directly, 11 of these were repealed before the end of the first year of the Protocol’s implementation and four were repealed during the second year.
Those repealed and replaced in 2021 concern:
-
- the approval and market surveillance of motor vehicles and related products (Directive 2007/46/EC) replaced by Regulation (EU) 2018/858 adopted in June 2018 and taking effect in August 2020;
- controls on cash entering or leaving the EU (Regulation (EC) 1889/2005) replaced by Regulation (EU) 2018/1672 adopted in November 2018 taking effect in June 2021;
- controls on trade in goods that could be used in capital punishment or torture (Council Regulation (EC) 1236/2005) replaced by Regulation (EU) 2019/125 adopted in January 2019 and taking effect in February 2019;
- the mutual recognition of goods between member states (Regulation (EC) 764/2008) replaced by Regulation (EU) 2019/515 adopted in March 2019 and taking effect in April 2020;
- controls on persistent organic pollutants (Regulation (EC) 850/2004) replaced by Regulation (EU) 2019/1021 adopted in June 2019 and taking effect in July 2019;
- the marketing and use of explosives precursors (Regulation (EU) 98/2013) replaced by Regulation (EU) 2019/1148 adopted in July 2019 and taking effect in January 2021;
- provisions for the conservation of fisheries and marine ecosystems (Council Regulation (EC) 850/98) replaced by Regulation (EU) 2019/1241 adopted in July 2019 taking effect in August 2019;
- provisions for computerising the movement and surveillance of exercisable goods (Decision 1152/2003/EC) replaced by Decision (EU) 2020/263 adopted in February 2020 and taking effect in March 2020;
- rules on the labelling of tyres (Regulation (EC) 1222/2009) replaced by Regulation 2020/740 adopted in June 2020 and taking effect in April 2021;
- controls on the acquisition and possession of weapons (Council Directive 91/447/EEC) replaced by Directive (EU) 2021/555 adopted and taking effect in April 2021; and
- the EU regime for the control of exports, transfer, brokering and transit of dual-use items (Regulation (EC) 428/2009) repealed by Regulation (EU) 2021/821 adopted and taking effect in May 2021 but with provision for the continued application of authorisations made under the earlier act and before 9 September 2021.
Those repealed and replaced in 2022 concern:
-
- the EU code relating to veterinary medicinal products (Directive 2001/82/EC) replaced by Regulation (EU) 2019/6 adopted in December 2018 and taking effect in January 2022;
- conditions governing the preparation, placing on the market and use of medicated feeding stuffs in the EU (Directive 90/167/EEC) replaced by Regulation (EU) 2019/4 adopted in December 2018 and taking effect January 2022;
- rules on the making available on the market of EU fertilising products (Regulation (EC) No 2003/2003) replaced by Regulation (EU) 2019/1009 adopted in June 2019 and taking effect in July 2022;
- regulations concerning certain categories of aid to related to the products, processing and marketing of fishery and aquaculture products (Commission Regulation (EU) No 1388/2014) replaced by Commission Regulation (EU) 2022/2473 adopted and taking effect in December 2022.
One additional act concerning plant protection (Council Directive 2000/29/EC) was repealed and replaced in December 2022 under provisions in Regulation (EU) 2016/2031 which already applies under the Protocol.
In addition to the 57 repealed acts, two acts originally listed in the Annexes expired after the UK withdrew from the EU. The expired acts concerned the regulation of imports from third countries affected by the Chernobyl disaster (Council Regulation (EC) 733/2008) and temporary trade measures for goods originating in Ukraine (Regulation (EU) 2017/1566).
Considering all these changes alongside those agreed to date by the Joint Committee, the number of EU acts that apply in post-Brexit Northern Ireland has decreased since the Protocol entered into force. As of 1 January 2023, there are now 308 EU regulations, directives and decisions that apply; 30 less than when the Protocol was first agreed in October 2019 (see Table 1). The decrease is essentially due, however, to the consolidation in a single replacement act of several previous acts and not a decrease in the scope of the Protocol.
4. Amendments to applicable EU law
A third category of change involves amendments to EU acts listed in the annexes to the Protocol. These are published as discrete pieces of legislation with ‘consolidated text’ versions of the Protocol-applicable EU act following.[2]
One of the few examples of this type of change is Commission Regulation ((EU) 2022/2046) introduced on 24 October 2022 which amends Regulation (EU) No 1408/2013 concerning the application of EU state aid rules in the agriculture sector. It replaced the maximum cumulative amounts that previously applied for the whole of the UK with ‘corresponding amounts’ for Northern Ireland alone. Regulation (EU) No 1408/2013 applies under Article 10 and Annex 5 of the Protocol. A consolidated text version of the regulation, published in November 2022, reflects the update.
Another example relates to the supply of medicines to Northern Ireland, a prominent issue in UK-EU talks on implementation of the Protocol. On 12 April 2022, the EU adopted Regulation (EU) 2022/641 to amend existing Regulation (EU) No 536/2014 concerning the regulation of medicines to introduce derogations from certain obligations for medicines that are available in Northern Ireland as well as in Ireland, Cyprus, and Malta. A consolidated text version of Regulation (EU) No 536/2014, which applies under Article 5 and Annex 2 of the Protocol, was published in December 2022, reflects the update.
5. Changes to EU legislation implementing applicable EU law
The fourth category of change relates to legislation that implements the EU acts listed in the Annexes to the Protocol. As with repeals, replacements, expiries and amendments, this type of change is the result of normal EU legislative processes. It also follows from Article 13(3) Protocol.
To understand the significance of this fourth category it is helpful to first explain what EU ‘implementing’ or ‘delegated’ legislation is and why it exists.
Often EU directives, regulations, and decisions, such as those listed in the Annexes to the Protocol, are written in quite general terms. They are, after all, designed to apply in all EU member states. This means, however, that the original ‘parent’ act – the piece of EU legislation listed in an Annex to the Protocol – does not always set out in detail all the procedures, processes or requirements that may be necessary to implement its provisions.
So, to avoid unhelpful ambiguity or unconstructive variation in the way a new law is implemented, EU acts often provide for implementing or delegated legislation to be adopted. The difference between implementing and delegated acts reflects the process for their adoption; the purpose of both is the same, namely, to implement the parent act. Such legislation is always within the scope of a given ‘parent’ act, sets out the rules and procedures for its operationalization, is adopted after the original parent act has been passed and according to its terms.
EU implementing legislation – including that applicable under the Protocol – is regularly adopted by either the Commission or the Council. In 2022, the EU adopted 1243 pieces of implementing legislation. Not all of these apply to Northern Ireland under the Protocol. Of the 1243 implementing acts adopted, 716 (representing 58%) were within the scope of the Protocol (see Table 2).
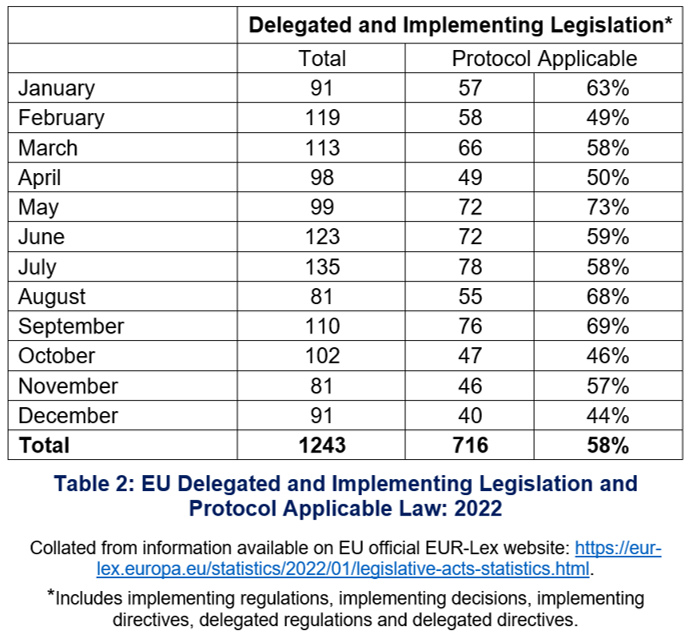
Figures for both total implementing acts adopted in 2022 and the proportion that are Protocol-applicable may seem high. It is important to note, however, that most implementing acts concern very technical, minor, and specific issues, and they remain within the scope of the original ‘parent’ act. Moreover, while all implementing acts made under ‘parent’ acts listed in the Protocol and its Annexes are applicable to Northern Ireland, not all of them are significant in terms of policy.
For example, implementing acts are adopted to correct errors in different language versions of an EU act: Commission Implementing Regulation (EU) 2022/176 made on 9 February 2022 corrects certain language versions of a particular Annex of the Official Controls Regulation (Regulation 2017/625) (noted above) and applies under Article 5 and Annex 2 of the Protocol; similarly Commission Implementing Regulation (EU) 2022/827 made on 20 May 2022 corrects the Danish language version of an Implementing Regulation ((EU) 2019/1842) that concerns arrangements for adjusting allocations of greenhouse gas emission allowances and applies under Article 9 and Annex 4 of the Protocol. While both of these implementing acts make changes to EU acts that apply to Northern Ireland under the Protocol, neither has an ‘on-the-ground’ impact.
Some technical changes are of significance, or potential significance, in and for Northern Ireland. For example, 72 of the EU implementing acts adopted in 2022 and which apply under the Protocol concern measures being taken across the EU and in Northern Ireland to address bird flu. While the primary purpose of these 72 implementing acts was technical – making amendments to lists of geographic regions where bird flu was or had been present – and many have already ceased to have effect, they also concern a very real issue facing the agrifood sector in Northern Ireland, and across the UK and EU so they are, in this respect, noteworthy.
Also, a small number of implementing acts that address Northern Ireland and its position under the Protocol directly have been adopted. On 21 February 2022, a Commission Implementing Regulation (EU) 2022/250 was made to amend existing EU implementing legislation to introduce a new model of animal health certificate for movements of certain livestock from Great Britain (GB) to Northern Ireland and so postpones the need for certificates regarding scrapie disease to be provided to allow time for GB holdings to be approved as ‘controlled risk’ despite being outside EU regulation.
On 27 April 2022, Commission Implementing Regulation (EU) 2022/680 was adopted to amend a standardized poster (provided for in Commission Implementing Regulation (EU) 2020/178 concerning the bringing of plants, fruits, vegetables, flowers or seeds, into the EU so as to include the ‘United Kingdom (Northern Ireland)’ in the list of non-EU territories for which there is an exemption from the ordinary requirement of a sanitary or phytosanitary (SPS) certificate for doing so. Again, the actual change here is very minor; however, it reflects the fact that the Protocol has provided for the continued free movement of goods on the island of Ireland, thereby negating the necessity for an SPS certificate that would otherwise be required in view of the UK no longer being in the EU.
While the examples above underline the often technical nature of provisions made in EU implementing legislation, they also demonstrate the potential for variation in terms of policy significance and sectoral impact in and for Northern Ireland. This is also why changes arising under the Protocol, including via implementing legislation, are important to track.
6. Tracking changes to applicable EU Law – EurLex… and Consolidated Texts
Tracking change, however, is not straightforward. Adopted EU legislation is published in the Official Journal of the European Union, but determining which pieces of EU law apply to Northern Ireland, and which do not, requires detailed study and timely cross-referencing.
A notable development in this respect was the launch in the summer of 2022 of webpages dedicated to EU law applicable under the Protocol. Established by the European Commission as part of the EUR-Lex website, these pages list all those EU acts that apply to Northern Ireland under the Protocol and so includes ‘parent’ acts, implementing acts, repealed acts, expired acts and replacement acts. From the UK government’s perspective, the EUR-Lex website reflects ‘the EU’s view of applicability’ with the Foreign Secretary stating that its content is ‘not endorsed’ by the UK government. Notwithstanding its ‘unilateral’ nature, as the only comprehensive official record of Protocol-applicable law, the EUR-Lex website is an important resource for anyone involved in, or affected by, the implementation of the Protocol. Calls have been made, notably by the House of Lords’ Protocol on Ireland/Northern Ireland Sub-Committee, for the UK government to produce its own ‘log’ or ‘audit’ of all EU legislation that applies in Northern Ireland. It has yet to do so.
Keeping track of applicable EU law under the Protocol is facilitated in part by the occasional publication of the ‘consolidated text’ versions of EU law. When a large number of amendments have been made to a ‘parent act’ of EU law (most often via EU implementing legislation) a ‘consolidated’ version of the legal text can be published in which all relevant changes are reflected. These consolidated versions of EU acts are produced for information purposes – they are not legal texts. Nonetheless, they bring together in one place changes and can provide a useful indicator of the extent to which those specific EU acts are evolving and are therefore worth monitoring.

EUR-Lex and the Applicable EU Law
(https://eur-lex.europa.eu/content/news/IENI.html)
Since 31 January 2020 when the UK left the EU consolidated text versions of 114 of those acts that apply under the Protocol have been published. Of these 95 were published after the end of the UK Transition Period on 1 January 2021.
7. Tracking changes to applicable EU Law – Post-Brexit Governance NI Research Database
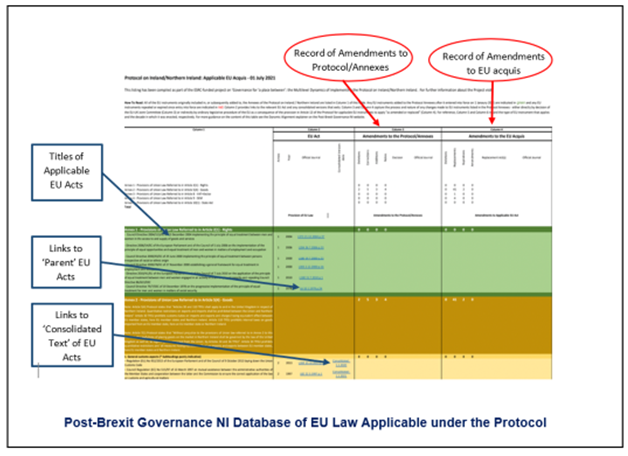
Tracking changes in EU law that apply under the Protocol is a particular focus of the three-year ESRC-funded research project Governance for ‘a place between’: the Multilevel Dynamics of Implementing the Protocol on Ireland/Northern Ireland for which this explainer has been produced.
To this end, the project has developed a freely accessible database that provides links to each applicable EU act as well as relevant implementing acts adopted by the EU that can be found through the ‘consolidated text’ versions of the original acts; any additions, amendments, or replacements as well as the deletions and instances where, through expiry, an EU act no longer applies are also recorded. The database contains a list of all of those EU acts that apply in the UK ‘in respect of Northern Ireland’ through the Protocol as of 1 January 2023. It therefore reflects all of the ‘amendments and replacements’ to applicable EU law described in this explainer.
Alongside this, the Post-Brexit Governance NI website maintains lists of relevant domestic UK laws that implement Protocol-applicable EU law where this is necessary. These include relevant Statutory Instruments passed in Westminster and Statutory Rules passed in Stormont. Examples include:
Statutory Instruments (Westminster)
-
-
- Human Medicines (Amendment) (Supply to Northern Ireland) Regulations 2021 (SI 2021/1452)
- Customs (Miscellaneous Provisions) (Amendment) (EU Exit) Regulations 2021 (SI 2021/1489)
- Customs (Amendment) (EU Exit) Regulations 2022 (SI 2022/109)
- Value Added (Enforcement Regulated to Distance Selling and Miscellaneous Amendments) Regulations 2022 (SI 2022/226).
-
Statutory Rules (Stormont)
-
-
- Edible Crabs (Conservation) (Amendment) Regulations (Northern Ireland) 2021 (SR 2021/336)
- Spring Traps Approval (Amendment) Order (Northern Ireland) 2021 (SR 2021/321)
- Official Controls (Plant Protection Products) Regulations (Northern Ireland) 2020 (SR 2020/360).
-
Reflecting the nature of ‘amendments and replacements' made at EU level, the content of these implementing UK laws tends to be technical and specific. To demonstrate, the Value Added Tax (Enforcement Regulated to Distance Selling and Miscellaneous Amendments) Regulations 2022 (SI 2022/226) is a statutory instrument made in March 2022 which corrects minor and consequential errors in domestic legislation related to simplified VAT accounting schemes (the ‘One Stop Shop’ and ‘Import One Stop Shop’) which form part of the EU e-commerce package that came into force in July 2021 and which the UK implements under the Protocol (see Explanatory Memorandum).
Conclusion: A dynamic democratic challenge
What is clear from this explainer is that implementation of the Protocol and the process of dynamic regulatory alignment involve extensive legislative complexity.
Since the terms of the UK’s withdrawal from the EU were agreed in October 2019, the EU acquis that applies under the Protocol has changed. To date, however, the majority of the most substantive ‘amendments and replacements’ enact changes agreed while the UK was still part of the EU.
This reflects the often-slow pace of EU legislative processes and the fact that the EU acquis that applies in post-Brexit Northern Ireland – primarily concerning trade in goods – is relatively stable. Moreover, many recent ‘amendments and replacements’ were made while the UK was an EU member state and so with the UK government’s agreement. That situation is now changing.
Moreover, as the UK increasingly diverges from what was EU law, and is currently retained EU law, the implications of Northern Ireland’s dynamic regulatory alignment can be expected to become more evident and more significant in respect to policy development and implementation.
This brings us to the challenges of practical impacts, democratic accountability, and legislative scrutiny posed by dynamic regulatory alignment for the maintenance of the conditions for the free movement of goods on the island of Ireland and the operation of the single electricity market. These challenges follow from both the novelty of the Protocol’s provisions and the degree of transparency (or lack thereof) surrounding the activities of the three bilateral UK-EU bodies set up to oversee their implementation: the Joint Committee, the Specialised Committee, and the Joint Consultative Working Group (JCWG).
At present, there is limited formal provision for those directly impacted by Northern Ireland’s position under the Protocol – Northern Ireland business, industry stakeholders, rights organisations, and community representatives – to input into the process of dynamic regulatory alignment and UK-EU discussions on their implications. This risks creating or exacerbating difficulties when it comes to the legitimacy of dynamic regulatory alignment and appreciation of the potential consequences of implementation
Dynamic regulatory alignment also has implications for the quality of democracy in Northern Ireland. Lack of provision for elected representatives in Northern Ireland to contribute to the development of Protocol-applicable EU laws or to scrutinise their adoption – as well as the development and adoption of amendments and implementing legislation – limits democratic accountability and transparency.
Notwithstanding these challenges, existing mechanisms, such as the JCWG, the Specialised Committee and the Joint Committee could be used to address and/or mitigate the difficulties, especially if proposals, such as those from the UK and EU and others are pursued and the institutional framework for the Protocol is enhanced. Consideration also needs to be given to how Northern Ireland interests can meaningfully inform the formal position of the UK government on any proposed EU legislative amendments and/or additions to EU laws made applicable by the Protocol.
Recent reports suggest that some form of agreement between the UK and EU on the implementation of the Protocol, potentially including some measures to improve Northern Ireland representation in and stakeholder engagement with the governance arrangements for the Protocol, may be forthcoming. Assuming such an agreement is reached, the new arrangements should contribute to improving capacity and opportunities to monitor the dynamic regulatory alignment with the body of EU law that applies in the UK in respect of Northern Ireland under the Protocol.
February 2023
Dr. Lisa Claire Whitten is Research Fellow on the Governance for ‘a place between’: the multilevel dynamics of implementing the Protocol on Ireland/Northern Ireland at Queen's University Belfast. She can be contacted via: l.whitten@qub.ac.uk.
Download a copy of this Explainer here
